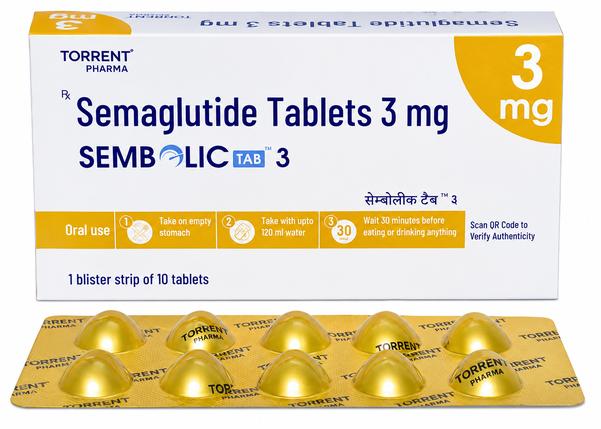
Sembolic 3 mg Tablet: The Premier Oral Initiation Therapy for Type 2 Diabetes Management Are you searching for a dependable Sembolic 3 mg tablet exporter from India? Ernest Impex, a division of the Group of Ernest Pharmaceutical Pvt. Ltd., is a leading international supplier of oral GLP-1 receptor agonists. We provide professional, high-integrity pharmaceutical distribution to Washington (USA), London (UK), Bangkok (Thailand), Canberra (Australia), Singapore, and Kingston (Jamaica). Extensive Product Profile: Understanding Sembolic 3 mg Sembolic 3 mg Tablet, manufactured by Torrent Pharmaceuticals Ltd., is an innovative oral formulation of Semaglutide. This 3 mg dose is specifically engineered as the initiation strength. Its primary purpose is to introduce the Semaglutide molecule to the body’s metabolic pathways gradually, reducing the risk of gastrointestinal side effects while beginning the process of glycemic optimization. 1. Clinical Indications & Usage Treatment Initiation: The mandatory first step for patients transitioning to oral GLP-1 therapy for Type 2 Diabetes. Glycemic Control: Used as an adjunct to diet and exercise to improve blood sugar (glucose) levels in adults with Type 2 Diabetes Mellitus. Complication Prevention: By stabilizing blood sugar, it helps reduce the long-term risk of kidney damage, nerve problems, and cardiovascular issues. Metabolic Preconditioning: Prepares the patient’s system for subsequent dose escalation to maintenance levels (7 mg and 14 mg). 2. Mechanism of Action: Complex Glucose Regulation Insulin Stimulation: It triggers the pancreas to release insulin in a glucose-dependent manner (only when blood sugar is high), preventing sudden drops. Glucagon Suppression: It inhibits the liver from secreting excess glucose into the bloodstream by lowering post-meal glucagon levels. Gastric Kinetic Delay: It slows the rate of stomach emptying, which results in more gradual sugar absorption and prolonged satiety (fullness). Appetite Modulation: It acts on specific receptors in the brain to reduce hunger and caloric intake, often leading to secondary weight management benefits. 3. Administration Protocol for Maximum Efficacy Empty Stomach: Must be taken immediately upon waking on an empty stomach. Strict Water Limit: Swallow the tablet whole with a small amount of plain water (maximum 120 ml / 4 ounces). The 30-Minute Window: Do not eat, drink, or take other medications for at least 30 minutes after ingestion. Storage, Handling, and Stability Protocols Primary Storage: Store in a cool, dry place at a temperature below 30°C. Moisture Barrier: Keep tablets in their original blister packaging until the moment of consumption. Exposure to atmospheric moisture can degrade the active molecule. Protection: Keep away from direct sunlight and heat. Do not store in bathrooms or near kitchen appliances where humidity levels fluctuate. PRESCRIPTION MEDICINE IS : YES Disclaimer: The information provided is for educational and informational purposes only and does not constitute professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare provider before making any medical or treatment decisions. 📞 Contact Ernest Impex — Pharmaceutical Exporter from India [GROUP OF ERNEST PHARMACEUTICAL PVT. LTD.] 🌐 Websites: www.ernestimpex.com www.ernestvision.com www.oncologymedicinesupplier.com www.ernestpharmaceuticals.com 📧 Email: exports@ernestpharmaceuticals.com 📦 Business Type: Exporter | Bulk Supplier | Distributor 📲 WhatsApp: +91 93599 02383 📲 WhatsApp Link: https://wa.me/919359902383