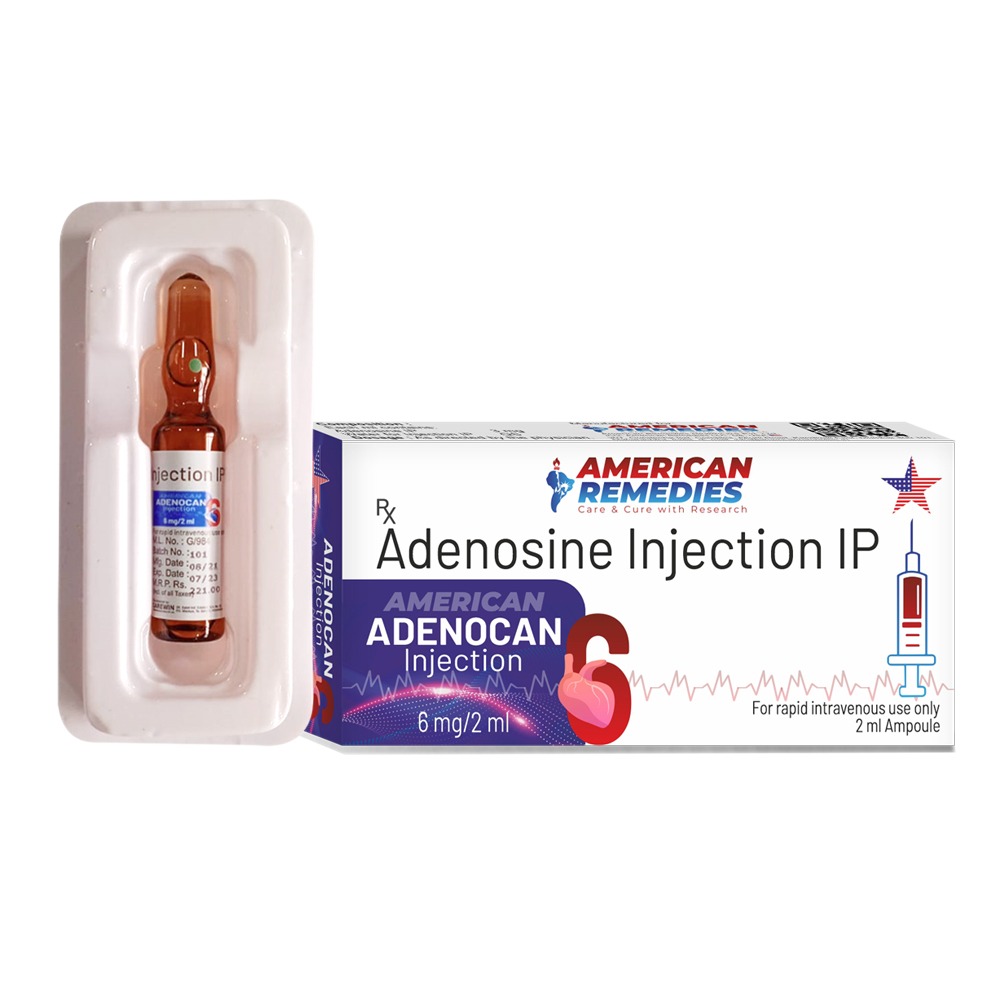
Adenoz 6mg Injection Marketer Celon Laboratories Ltd SALT COMPOSITION Adenosine (6mg) Storage Store below 25°C Product introduction Adenoz 6mg Injection is an anti-arrhythmic medicine used to treat life-threatening arrhythmia (abnormal heart rhythm). It restores normal heart rhythm by blocking abnormal electrical signals in the heart. Adenoz 6mg Injection is also used to carry out diagnostic stress tests of the heart. It is administered under the supervision of a doctor. Your doctor will decide the dose and duration of the medicine for you. The doctor may closely monitor your blood pressure, heart rate, and ECG until you become stable. You should continue all your other medicines that are advised by the doctor for your long-term treatment even after this injection. Common side effects of this medicine include flushing (sense of warmth in the face, ears, neck, and trunk), headache, chest pain, dizziness, and nausea. Consult your doctor if these effects bother you. This medicine is used with caution for some people. Thus, you must inform the doctor if you are suffering from any kidney or liver disease. Also, let your doctor know if you are pregnant or breastfeeding and about all the other medications that you are taking regularly. Uses of Adenoz Injection Arrhythmia Side effects of Adenoz Injection Most side effects do not require any medical attention and disappear as your body adjusts to the medicine. Consult your doctor if they persist or if you’re worried about them Common side effects of Adenoz Flushing (sense of warmth in the face, ears, neck and trunk) Dizziness Lightheadedness Nausea Headache Numbness Chest pain How to use Adenoz Injection Your doctor or nurse will give you this medicine. Kindly do not self administer. How Adenoz Injection works Adenoz 6mg Injection is an anti-arrhythmic medication. It regulates heart beat by blocking abnormal electrical signals in the heart. Safety advice warnings Alcohol CONSULT YOUR DOCTOR It is not known whether it is safe to consume alcohol with Adenoz 6mg Injection. Please consult your doctor. warnings Pregnancy CONSULT YOUR DOCTOR Adenoz 6mg Injection may be unsafe to use during pregnancy. Although there are limited studies in humans, animal studies have shown harmful effects on the developing baby. Your doctor will weigh the benefits and any potential risks before prescribing it to you. Please consult your doctor. warnings Breast feeding SAFE IF PRESCRIBED Adenoz 6mg Injection is probably safe to use during breastfeeding. Limited human data suggests that the drug does not represent any significant risk to the baby. warnings Driving NOT RELEVANT Not relevant, as Adenoz 6mg Injection is intended for use in hospitalised patients. warnings Kidney SAFE IF PRESCRIBED Adenoz 6mg Injection is safe to use in patients with kidney disease. No dose adjustment of Adenoz 6mg Injection is recommended. warnings Liver SAFE IF PRESCRIBED Adenoz 6mg Injection is probably safe to use in patients with liver disease. Limited data available suggests that dose adjustment of Adenoz 6mg Injection may not be needed in these patients. Please consult your doctor.