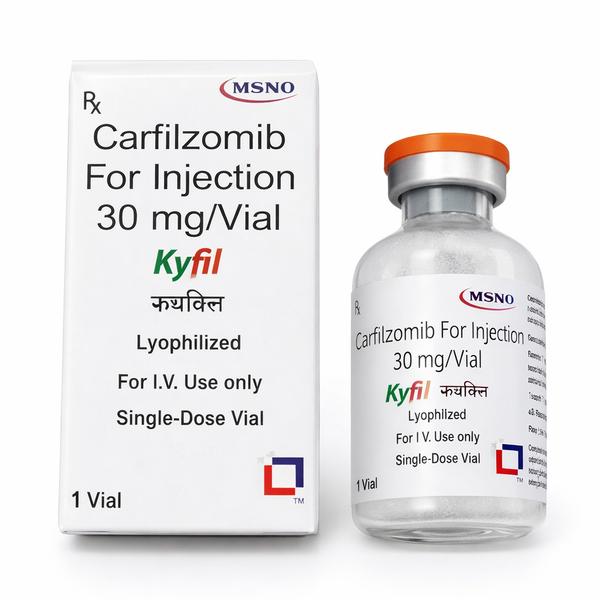
Kyfil 30 mg Injection: Targeted Second-Generation Proteasome Inhibitor for Relapsed and Refractory Multiple Myeloma Therapeutics Extensive Product Profile: In-Depth Insights on Kyfil 30 mg Kyfil 30 mg Injection, manufactured under strict global regulatory quality standards by MSN Laboratories, contains Carfilzomib as its active pharmaceutical ingredient. Supplied as a sterile, single-dose lyophilized powder inside an injection vial, it represents a core therapeutic protocol designed to overcome clinical resistance seen with first-generation proteasome treatments. 1. Clinical Indications & Target Therapeutic Application Relapsed or Refractory Multiple Myeloma (RRMM): Formulated for patients whose bone marrow plasma cell cancer has progressed or returned after receiving at least one to three prior anti-cancer therapies. Synergistic Combination Infusion Regimens: Extensively used in combination therapies with immunomodulatory medicines (like Lenalidomide) and corticosteroids (like Dexamethasone) to significantly increase progression-free survival (PFS) rates. Monotherapy Protocols: Administered as a standalone option for patients who show resistance to both bortezomib and immunomodulatory agents. 2. Advanced Mechanism of Action (MOA) Irreversible 20S Proteasome Targeting: Carfilzomib is a selective, epoxyketone-class proteasome inhibitor. It binds covalently and irreversibly to the chymotrypsin-like active sites of the 20S proteasome core contained inside the larger 26S proteasome complex. Inhibition of Waste Degradation: By blocking this crucial enzymatic breakdown pathway, Kyfil halts the normal recycling of regulatory, misfolded, and polyubiquitinated proteins. Toxic Protein Build-up and Apoptosis: Because multiple myeloma cells reproduce rapidly and over-produce abnormal proteins, they depend heavily on functional proteasomes to survive. When Kyfil stops this mechanism, a rapid, toxic backlog of proteins accumulates inside the cell. 3. Cold-Chain Storage, Handling, and Stability Protocols Refrigeration Matrix: Prior to reconstitution, unopen vials must be stored and transported strictly within a validated cold chain framework between 2°C to 8°C (36°F to 46°F). Do not freeze. Light Mitigation: Retain the vial safely inside its original outer cardboard carton to shield the lyophilized compound from ambient light-induced degradation. Post-Reconstitution Storing Window: Once mixed with Sterile Water for Injection, the solution must be administered immediately. Reconstituted vials can be kept refrigerated at $2^circtext{C}$ to $8^circtext{C}$ for up to 24 hours, or kept at room temperature ($15^circtext{C}$ to $30^circtext{C}$) for no longer than 4 hours before infusion. Potential Side Effects of Kyfil 30 mg Hematological Complications: Pronounced reduction in red blood cells (Anemia), blood platelets (Thrombocytopenia), and white blood cells (Neutropenia/Leukopenia). This significantly elevates risks of fatigue, bruising, and severe infections. Regular Complete Blood Counts (CBC) are mandatory. Cardiovascular & Pulmonary Toxicities: Shortness of breath (Dyspnea), sudden spikes in blood pressure (Hypertension), fluid retention (Peripheral Edema), and rare but serious risks of congestive heart failure. Gastrointestinal Disturbances: Chronic nausea, diarrhea, vomiting, stomach pain, and decreased appetite. Infusion Site Reactions: Pyrexia (fever), chills, joint or muscle pain, and flushing occurring within 24 hours of administration. Renal Impact: Risk of acute kidney injury or tumor lysis syndrome; keeping the patient properly hydrated before and after the infusion is essential. Export Documentation & Regulatory Compliance Certificate of Analysis (COA): Offering independent laboratory verification confirming the exact 30 mg strength, chemical purity, and absolute sterility of the Carfilzomib batch. Certificate of Origin (COO): Authenticating that the product has been processed in a registered, state-of-the-art GMP-certified manufacturing facility in India Validated Cold-Chain Logs: Continuous data-logger readings to prove the maintenance of mandatory cold chain temperatures during global air transit. PRESCRIPTION MEDICINE IS YES Disclaimer: This product profile is generated for international business procurement, trade distribution, and B2B informational awareness only. Kyfil (Carfilzomib) is a high-potency, restricted prescription chemotherapy injection that must be prepared, handled, and administered exclusively by qualified healthcare providers within a licensed medical facility or clinical setting. 📞 Contact Ernest Impex — Pharmaceutical Exporter from India [GROUP OF ERNEST PHARMACEUTICAL PVT. LTD.] 🌐 Websites: www.ernestimpex.com www.ernestvision.com www.oncologymedicinesupplier.com www.ernestpharmaceuticals.com 📧 Email: exports@ernestpharmaceuticals.com 📦 Business Type: Exporter | Bulk Supplier | Distributor 📲 WhatsApp: +91 93599 02383 📲 WhatsApp Link: https://wa.me/919359902383