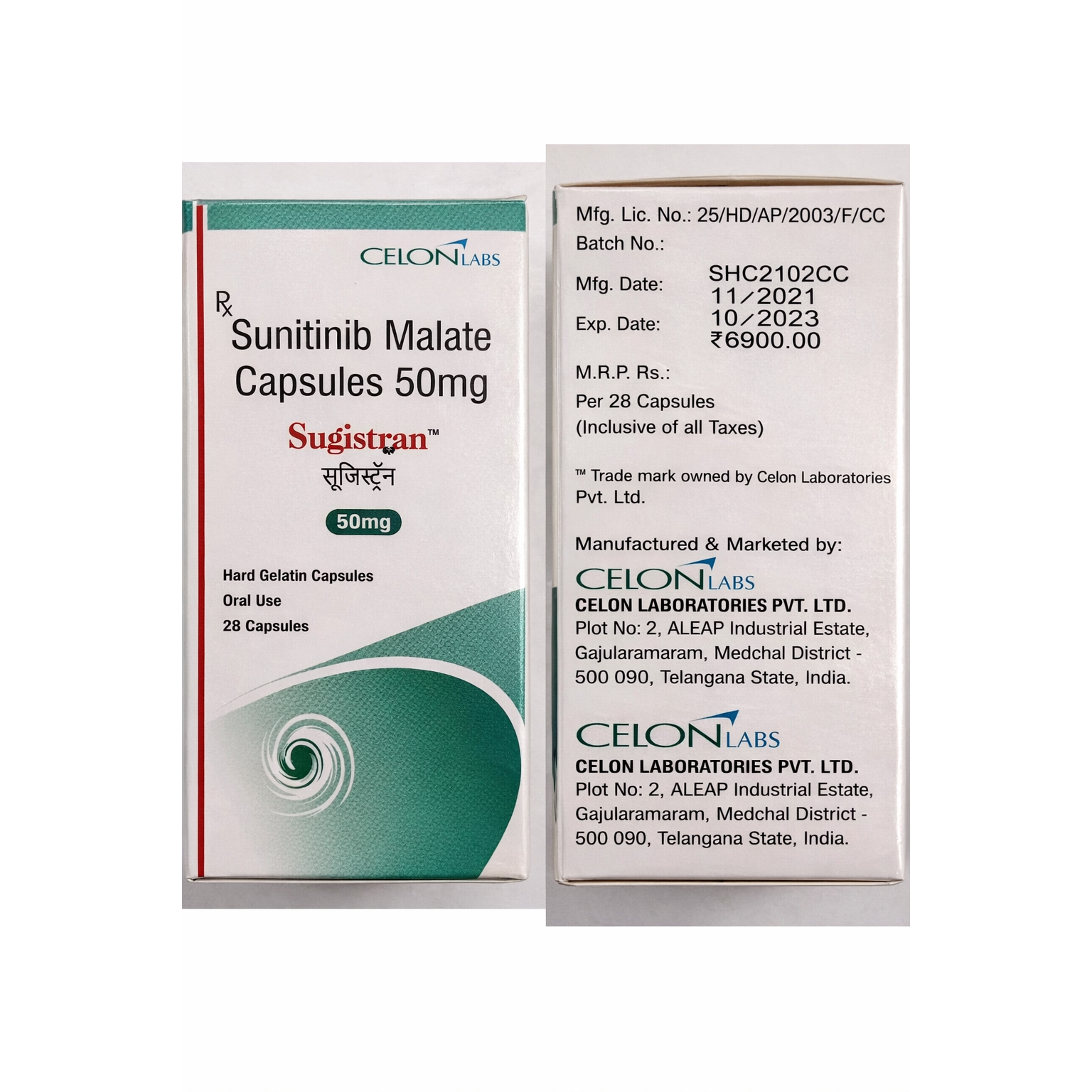
Sugistran 50mg Capsule: Precision Multi-Kinase Inhibition for Targeted Oncology Excellence
Are you seeking a reliable Sugistran 50 mg capsule exporter from India? Ernest Impex, a specialized division of the Group of Ernest Pharmaceutical Pvt. Ltd., is a premier global supplier and bulk distributor of targeted anti-cancer agents. We facilitate a regulatory-compliant, cold-chain-integrated supply chain serving Washington (USA), London (UK), Bangkok (Thailand), Canberra (Australia), Singapore, and Kingston (Jamaica), ensuring that critical oncology therapies are delivered with maximum clinical efficacy.
Detailed Product Overview: What is Sugistran 50 mg?
Sugistran 50mg Capsule is an oral multi-targeted tyrosine kinase inhibitor (TKI) containing the active ingredient Sunitinib.
For international B2B procurement, Sugistran 50mg is a gold-standard choice for treating advanced solid tumors that require powerful, multi-pathway suppression.
Primary Therapeutic Indications
Sugistran 50 mg is clinically utilized globally for:
Metastatic Renal Cell Carcinoma (mRCC): A primary treatment for advanced stage kidney cancer.
Gastrointestinal Stromal Tumor (GIST): Specifically for rare tumors in the digestive tract that are resistant to or cannot be treated with Imatinib.
Pancreatic Neuroendocrine Tumors (pNET): For progressive, well-differentiated tumors located in the pancreas.
Mechanism of Action: Multi-Pathway Signal Inhibition
Sugistran 50mg operates through a sophisticated dual-action process that targets both tumor growth and the tumor's life-support system:
Receptor Blockade: It inhibits multiple receptor tyrosine kinases (RTKs), specifically targeting vascular endothelial growth factor receptors (VEGFR-1, -2, -3) and platelet-derived growth factor receptors (PDGFR-α, -β).
Anti-Angiogenesis: By blocking the VEGFR pathway, Sugistran prevents the tumor from developing the new blood vessels it needs to "feed" its growth.
Direct Growth Arrest: It targets KIT and FLT3 receptors, which are vital for the uncontrolled division of cancer cells in gastrointestinal and hematological malignancies.
Systemic Starvation: This combined action starves the tumor of nutrients while simultaneously halting the internal signals for cell division.
Administration and Clinical Safety
Dosage Schedule: Typically taken once daily.
For GIST and RCC, the standard protocol is often 4 weeks on treatment followed by a 2-week break. Storage: Store at room temperature (below 30°C) in a dry place.
Keep the container tightly closed to protect from light. Key Clinical Monitoring:
Blood Pressure: Hypertension is a common side effect; regular monitoring is mandatory.
Thyroid Function: Routine checks for hypothyroidism are standard during Sunitinib therapy.
Liver & Cardiac Health: Periodic liver function tests (LFTs) and cardiovascular assessments (LVEF) are recommended.
Blood Sugar: Diabetics should monitor blood glucose levels closely, as Sugistran can cause hypoglycemia.
Why Source Sugistran from Ernest Impex?
Authentic Celon Oncology Exporter: Direct access to genuine Celon Laboratories inventory, ensuring 100% product authenticity and competitive bulk pricing.
Specialized Logistics Authority: We specialize in the complex documentation and cold-chain logistics required for Washington, London, Bangkok, Canberra, Singapore, and Kingston.
Regulatory Transparency: We provide all essential B2B documentation, including GMP compliance, batch-specific COAs, and necessary export permits.
Bulk Supply Stability: We specialize in large-scale fulfillment for hospital pharmacies, specialty oncology centers, and regional distributors.
📞 Contact Ernest Impex — Pharmaceutical Exporter from India
[GROUP OF ERNEST PHARMACEUTICAL PVT. LTD.]
🌐 Websites:
📧 Email:
📦 Business Type: Exporter | Bulk Supplier | Distributor
📲 WhatsApp: +91 93599 02383