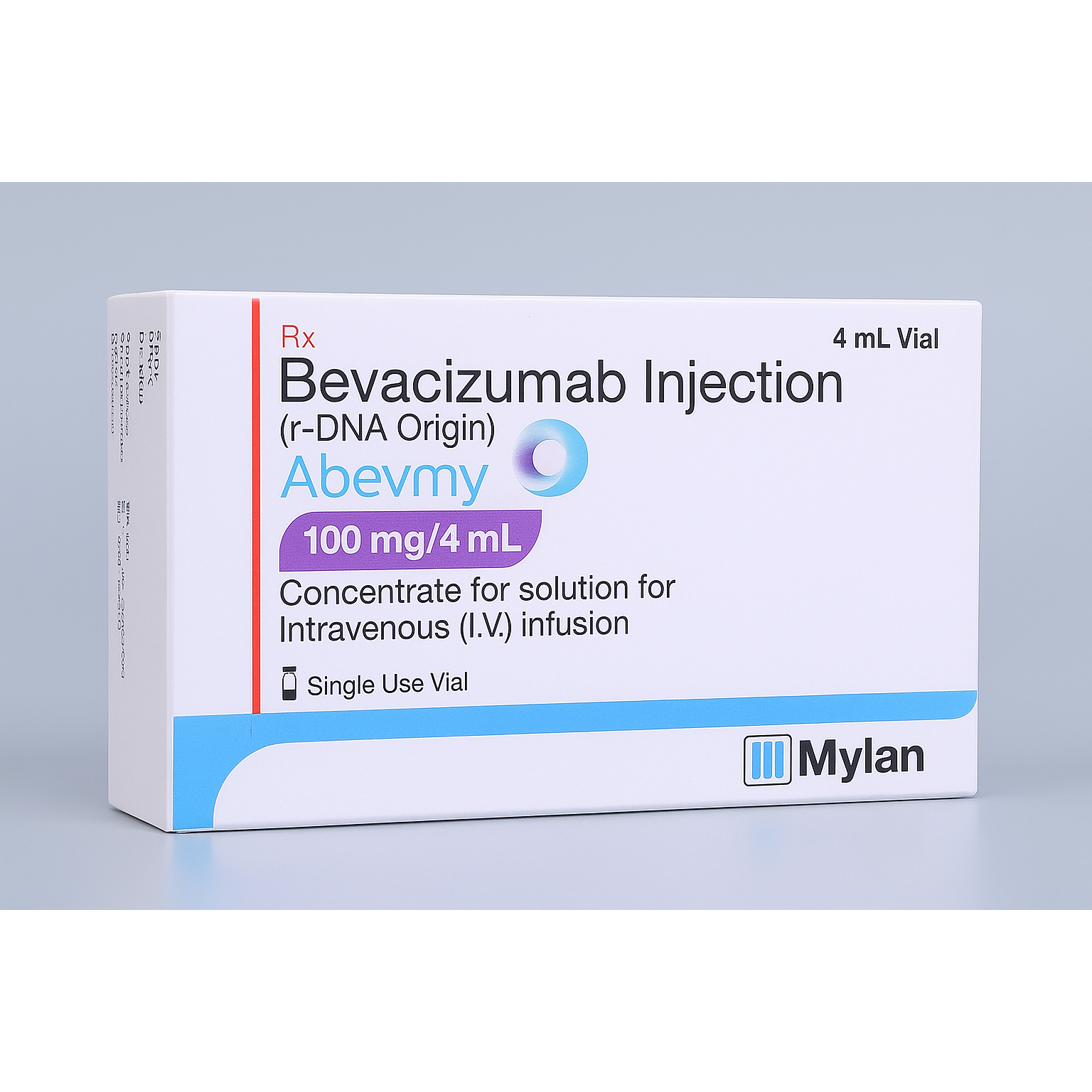
Abevmy 100 Solution for Infusion (4 ml)
United States (Washington D.C., New York City, Los Angeles, Chicago, Houston, Phoenix)
Composition: Bevacizumab 100 mg
Manufacturer / Marketer: Mylan Pharmaceuticals Pvt Ltd
Dosage Form: Parenteral (for intravenous infusion)
Storage: Store under cold-chain conditions at 2°C – 8°C. Protect from light.
Product Overview
Abevmy 100 Solution for Infusion 4 ml is a monoclonal antibody-based anticancer medicine containing Bevacizumab as the active ingredient. It is prescribed for the management of several solid tumors including metastatic breast cancer, colorectal cancer, non-small cell lung cancer, renal cell carcinoma, cervical cancer, epithelial ovarian cancer, fallopian tube cancer, primary peritoneal cancer, glioblastoma, and hepatocellular carcinoma.
Abevmy acts by inhibiting Vascular Endothelial Growth Factor (VEGF) — a protein that promotes the growth of blood vessels supplying nutrients to tumors. By blocking VEGF, the medicine helps reduce tumor blood supply, thereby slowing or stopping tumor growth.
@ERNESTPHARMACEUTICALPVTLTD
For bulk quotations, availability, and orders, please contact us:
📧 Email: exports@ernestpharmaceuticals.com
🌐 www.ernestpharmaceuticals.com| www.ernestimpex.com
Amit Rai: WhatsApp link https://wa.me/message/M6GESWXSIOZMD1
📲 WhatsApp: +91 94054 76170
Therapeutic Indications
Metastatic Breast Cancer
Colorectal Cancer (in combination with chemotherapy)
Non-Small Cell Lung Cancer (NSCLC)
Renal Cell Carcinoma (Kidney Cancer)
Cervical, Ovarian, Fallopian Tube, and Peritoneal Cancer
Glioblastoma (Brain Tumor)
Hepatocellular Carcinoma (Liver Cancer)
Mechanism of Action
Bevacizumab, the active component in Abevmy, binds specifically to VEGF, preventing its interaction with VEGF receptors on the surface of endothelial cells. This action inhibits angiogenesis — the formation of new blood vessels — depriving cancerous tissues of oxygen and nutrients necessary for their growth and metastasis.
Administration & Dosage
Abevmy 100 Solution for Infusion is administered intravenously by a qualified healthcare professional in a hospital or clinical setting. The dosage and treatment schedule depend on the patient’s clinical condition and the type of cancer being treated. Self-administration is not recommended.
Safety & Precautions
Not recommended during pregnancy or breastfeeding.
Effective contraception should be used during treatment and for 6 months after the last dose.
May cause dizziness or drowsiness — avoid driving or operating machinery after infusion.
Regular medical monitoring is advised to assess response and detect side effects.
Possible Side Effects
Common side effects may include fatigue, fever, loss of appetite, constipation, nasal bleeding, changes in taste, or mild speech disturbances. Most reactions are temporary; however, persistent or severe effects should be reported to a healthcare provider immediately.
Storage & Handling
Store under refrigerated conditions between 2°C and 8°C.
Do not freeze or shake.
Keep away from direct sunlight and out of reach of children.