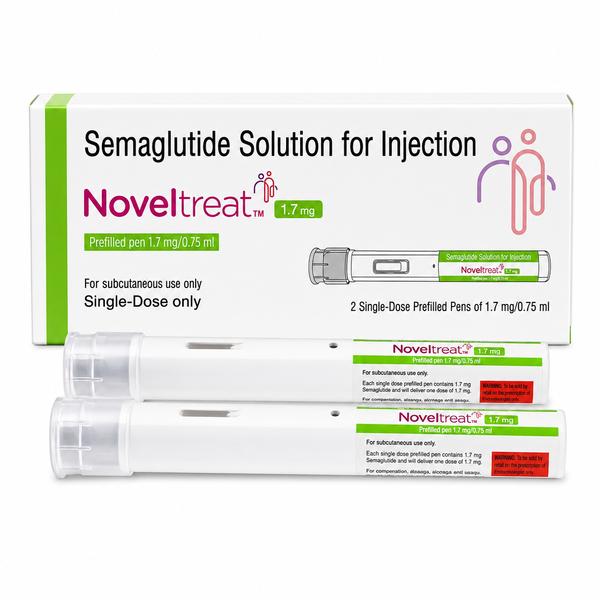
Noveltreat 1.7 mg Pre-filled Pen: High-Potency Maintenance Therapy for Weight Loss Are you seeking a trusted Noveltreat 1.7 mg Pre-filled Pen exporter from India? Ernest Impex, a key member of the Group of Ernest Pharmaceutical Pvt. Ltd., is a global specialist in the wholesale distribution of advanced GLP-1 receptor agonists. We provide robust, GDP-compliant supply chains and secure pharmaceutical logistics to Washington (USA), London (UK), Bangkok (Thailand), Canberra (Australia), Singapore, and Kingston (Jamaica). Extensive Product Profile: Understanding Noveltreat 1.7 mg Noveltreat 1.7 mg Pre-filled Pen contains Semaglutide, a potent metabolic regulator manufactured by Sun Pharmaceutical Industries Ltd. As an advanced maintenance strength, the 1.7 mg dose is designed for patients who require intensive support for long-term weight maintenance and cardiovascular risk reduction after completing lower-dose titration phases. 1. Clinical Indications & Usage Chronic Weight Management: Indicated for long-term weight loss and maintenance in adults with obesity (BMI $ge$ 30 $kg/m^2$) or overweight (BMI $ge$ 27 $kg/m^2$) with weight-related comorbidities. Adolescent Obesity: Specifically approved for adolescents aged 12 years and older with a high initial BMI. Cardiovascular Protection: Clinically used to reduce the risk of major adverse cardiovascular events (MACE) such as heart attack, stroke, or death in adults with established heart disease and high BMI. Liver Health: Emerging evidence supports its use in treating non-cirrhotic non-alcoholic steatohepatitis (NASH) with moderate to advanced liver scarring. 2. Mechanism of Action: The GLP-1 Pathway Appetite Regulation: It mimics a natural hormone that targets the hypothalamus in the brain, increasing feelings of fullness and significantly curbing food cravings. Slowed Digestion: It slows down gastric emptying, leading to prolonged satiety and more stable blood sugar levels after meals. Insulin Response: It stimulates the pancreas to release insulin in a glucose-dependent manner, while simultaneously reducing the liver's production of excess sugar. 3. Administration and Dosing Dose: Injected once weekly on the same day each week. Escalation Path: This 1.7 mg strength is typically reached after sequential 4-week periods on 0.25 mg, 0.5 mg, and 1.0 mg doses. Route: Subcutaneous injection in the abdomen, thigh, or upper arm. Storage, Handling, and Stability Protocols Primary Storage: Unused pens must be stored in a refrigerator between $2^circtext{C}$ and $8^circtext{C}$. Stability After Use: Once the pen is first used, it can be kept at room temperature (below $30^circtext{C}$) or in the refrigerator for up to 56 days. Light Protection: Keep the pen cap on to shield the medication from light exposure.Safety Warning: Do not freeze. If the solution becomes frozen, it is denatured and must be discarded. Documentation & Global Export Compliance Commercial Invoice Certificate of Analysis (COA) Bill of Lading / Airway Bill Export License & Regulatory Support: We facilitate document clearance for TGA, HSA, US-FDA, and MHRA import requirements. PRESCRIPTION MEDICINE IS: YES Disclaimer: The information provided is for educational and informational purposes only and does not constitute professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare provider before making any medical or treatment decisions. 📞 Contact Ernest Impex — Pharmaceutical Exporter from India [GROUP OF ERNEST PHARMACEUTICAL PVT. LTD.] 🌐 Websites: www.ernestimpex.com www.ernestvision.com www.oncologymedicinesupplier.com www.ernestpharmaceuticals.com 📧 Email: exports@ernestpharmaceuticals.com 📦 Business Type: Exporter | Bulk Supplier | Distributor 📲 WhatsApp: +91 93599 02383 📲 WhatsApp Link: https://wa.me/919359902383