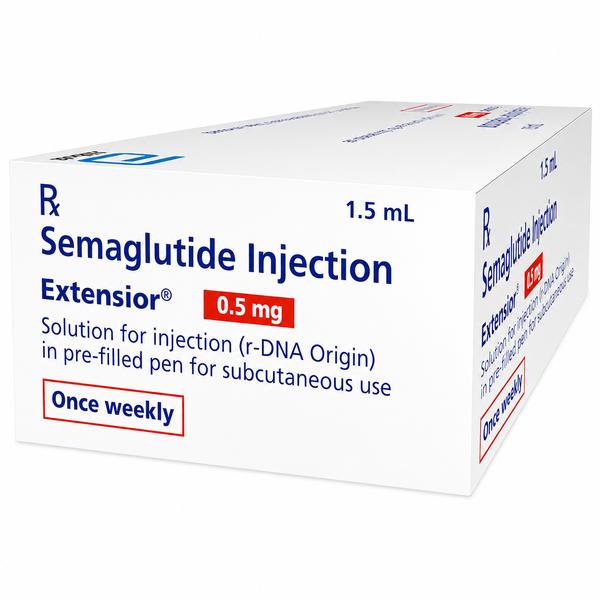
Extensior 0.5 mg Injection Combipack: Sustainable Maintenance for Metabolic and Weight Wellness Are you searching for a certified Extensior 0.5 mg injection combipack exporter from India? Ernest Impex, a key global division of the Group of Ernest Pharmaceutical Pvt. Ltd., is a premier supplier of high-demand metabolic and endocrine formulations. We provide specialized, cold-chain logistics and secure bulk supply to Washington (USA), London (UK), Bangkok (Thailand), Canberra (Australia), Singapore, and Kingston (Jamaica). Extensive Product Profile: Understanding Extensior 0.5 mg Extensior 0.5 mg Injection Combipack, manufactured by Abbott India Ltd., contains the active ingredient semaglutide. This 0.5 mg strength serves as a vital step in the treatment titration protocol. Following the 0.25 mg initiation phase, the 0.5 mg dose is designed to provide enhanced glycemic control and sustainable weight management while the body continues to adjust to the GLP-1 hormone. 1. Clinical Indications & Usage (2026 Guidelines) Chronic Weight Management: Indicated for long-term weight reduction and maintenance in adults with obesity ($BMI ge 30 kg/m^2$) or overweight ($BMI ge 27 kg/m^2$) who have at least one weight-related condition such as hypertension or dyslipidemia. Cardiovascular Risk Reduction: Clinically proven to lower the risk of major adverse cardiovascular events (MACE), including heart attack, stroke, or cardiovascular death, in adults with established heart disease. Type 2 Diabetes Management: Used as an adjunct to diet and exercise to improve blood sugar control in adults. Adolescent Metabolic Health: Approved for chronic weight management in pediatric patients aged 12 years and older who meet specific obesity thresholds. 2. Mechanism of Action: The Science of Metabolic Signaling Appetite Modulation: It acts on the brain’s hypothalamus to increase feelings of satiety (fullness) and significantly reduce food cravings. Glucoregulatory Control: It stimulates the pancreas to release insulin only when blood sugar levels are high and inhibits the liver from releasing unnecessary sugar. Gastric Kinetic Delay: It slows the rate at which food leaves the stomach, which leads to more gradual sugar absorption and prolonged post-meal satisfaction. 3. The “Combipack“ Advantage One Pre-filled Pen (PFP): Containing 1.5 ml of solution (precalibrated for 0.5 mg doses). Four Sterile Disposable Needles: Providing a complete 4-week supply for a full month of therapy. Storage, Handling, and Stability Protocols Primary Cold Chain: Store unused pens in a refrigerator between 2°C and 8°C. Stability After Use: Once the pen is first used, it can be kept at controlled room temperature (up to 30°C) or in the refrigerator for up to 56 days. Light Protection: Always keep the pen cap on to protect the solution from light degradation. Crucial Warning: Do not freeze. Freezing permanently damages the peptide structure. If the solution becomes frozen, it must be discarded immediately. PRESCRIPTION MEDICINE IS: YES. Disclaimer: The information provided is for educational and informational purposes only and does not constitute professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare provider before making any medical or treatment decisions. 📞 Contact Ernest Impex — Pharmaceutical Exporter from India [GROUP OF ERNEST PHARMACEUTICAL PVT. LTD.] 🌐 Websites: www.ernestimpex.com www.ernestvision.com www.oncologymedicinesupplier.com www.ernestpharmaceuticals.com 📧 Email: exports@ernestpharmaceuticals.com 📦 Business Type: Exporter | Bulk Supplier | Distributor 📲 WhatsApp: +91 93599 02383 📲 WhatsApp Link: https://wa.me/919359902383