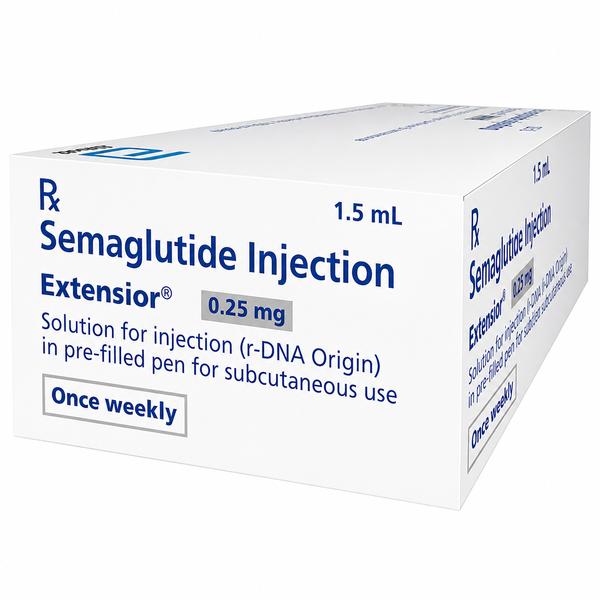
Extensior 0.25 mg Injection Combipack: The Essential Foundation for GLP-1 Therapy Initiation Are you looking for a trusted Extensior 0.25 mg injection combipack exporter from India? Ernest Impex, a premier division of the Group of Ernest Pharmaceutical Pvt. Ltd., is a global leader in the distribution of advanced metabolic treatments. We specialize in secure, cold-chain pharmaceutical logistics for initiation-phase GLP-1 therapies, serving medical professionals and bulk buyers in Washington (USA), London (UK), Bangkok (Thailand), Canberra (Australia), Singapore, and Kingston (Jamaica). Extensive Product Profile: Understanding Extensior 0.25 mg Extensior 0.25 mg Injection Combipack is a high-precision pharmaceutical product manufactured by Abbott India Ltd. Containing the active ingredient Semaglutide, this Combipack is specifically designed as the “Step 1“ initiation dose. It is engineered to gradually introduce the Semaglutide molecule to the body’s receptors, minimizing gastrointestinal sensitivity while laying the groundwork for effective long-term weight and glucose management. 1. Clinical Indications & Primary Usage Treatment Initiation: The mandatory starting dose for patients beginning chronic weight management or Type 2 Diabetes therapy. Chronic Weight Management: Indicated as an adjunct to a reduced-calorie diet and increased physical activity for adults with a BMI $ge$ 30 $kg/m^2$ or $ge$ 27 $kg/m^2$ with weight-related comorbidities. Cardiovascular Risk Reduction: Utilized to reduce the risk of major adverse cardiovascular events (heart attack, stroke) in adults with established heart disease. Metabolic Stabilization: Helps in improving HbA1c levels and stabilizing insulin sensitivity from the very first month of treatment. 2. Mechanism of Action: The Science of Satiety and Control Extensior functions as a selective GLP-1 (Glucagon-Like Peptide-1) receptor agonist. Its mechanism is a multi-pathway biological response: Hypothalamic Appetite Suppression: It targets the areas of the brain responsible for hunger, enhancing the sensation of fullness and reducing food cravings. Glucoregulatory Dynamics: It stimulates insulin secretion in a glucose-dependent manner (only when blood sugar is high) and inhibits the inappropriate release of glucagon. Gastric Kinetic Delay: By slowing down gastric emptying, it ensures nutrients are processed more slowly, leading to prolonged satiety and more stable post-meal glucose levels. 3. The “Combipack“ Advantage The Extensior 0.25 mg Combipack is designed for patient compliance and convenience. Each pack typically includes: One Pre-filled Pen (PFP): Containing 1.5 ml of solution (delivering four 0.25 mg doses). Four Sterile Disposable Needles: Ensuring the patient has a complete supply for the first month of treatment. Storage, Handling, and Stability Protocols Primary Cold-Chain: Store unused pens in a refrigerator at 2°C to 8°C. In-Use Period: Once the pen is first used, it can be kept at controlled room temperature (up to 30°C) or in the refrigerator for up to 56 days. Light Sensitivity: Always keep the pen cap on when not in use to protect the medicine from light. Safety Critical: Do not freeze. If the medication is accidentally frozen, it must be discarded immediately. PRESCRIPTION MEDICINE IS :YES Disclaimer: The information provided is for educational and informational purposes only and does not constitute professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare provider before making any medical or treatment decisions. 📞 Contact Ernest Impex — Pharmaceutical Exporter from India [GROUP OF ERNEST PHARMACEUTICAL PVT. LTD.] 🌐 Websites: www.ernestimpex.com www.ernestvision.com www.oncologymedicinesupplier.com www.ernestpharmaceuticals.com 📧 Email: exports@ernestpharmaceuticals.com 📦 Business Type: Exporter | Bulk Supplier | Distributor 📲 WhatsApp: +91 93599 02383 📲 WhatsApp Link: https://wa.me/919359902383